Water Quality Impacts Pesticide Performance
Grateful for the rain! With the anticipation of many post-herbicide applications occurring this week, just a reminder to make sure you know the specific herbicide traits each field has to avoid incorrect products applied and potential crop damage. The remainder of this column will be about the importance of water quality in pesticide applications, but is a brief overview. Additional resources are mentioned at the end. Water quality including pH, hardness, bicarbonate, and turbidity can make a difference on the efficacy of the pesticide. One always needs to read the pesticide label to determine what pH is best for the spray solution and also any other factors it mentions about water quality for the most effective pesticide application.
For example, do you check the pH of the final pesticide solution before it gets sprayed on the field? As a quick reminder, a pH of 7.0 is neutral. Above that is alkaline (basic) and less than 7.0 is acidic. Many of us have well water that is more alkaline. For example, my well water has a pH of 8.0. In general, herbicides, insecticides, and fungicides perform best in slightly acidic water (if the label doesn’t mention a specific pH assume between a pH of 5.5-6.5). Many herbicides are formulated as weak acids. Reading the pesticide label is important, though, because some pesticides like sulfonyurea herbicides and copper fungicides need slightly alkaline solutions.
Why is the pH important? The pH of the pesticide solution can impact the product performance including rapidly reducing the pesticide half-life. The half-life of a pesticide is the time it takes for that product’s active ingredient to degrade 50% in the environment. For example, a herbicide’s half life can go from just 10 minutes in a pH solution of 9.0 to 16 days at a pH solution of a 5.0. That’s one of the reasons why one should check the pH of the water using a pH meter before a buffering agent and any pesticide is added to the tank. It’s good to test the pH as various products are added to the tank and before the final solution is sprayed onto the field.

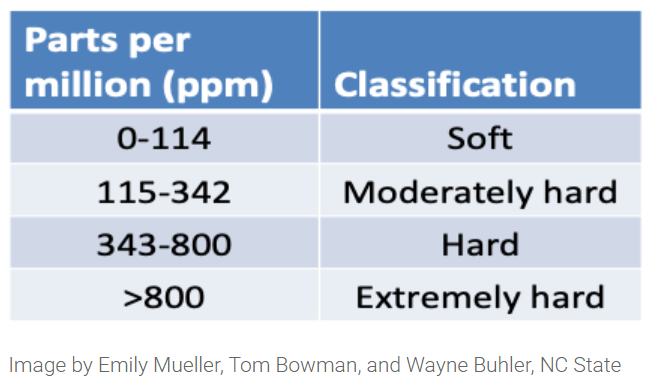
Water hardness can also impact pesticide performance by creating a poor mix, not allowing the product to dissolve in water, or reducing the pesticide half-life. Water hardness mostly indicates the amount of calcium and magnesium in the water, but aluminum, iron, sodium, and potassium are also positive ions that can bind to the pesticide potentially forming precipitates. Water hardness should in general be below 342 ppm. Water quality can be tested by sending a water sample to a laboratory.
The following are some corrective measures from North Carolina State University that can be taken to help correct poor water quality in pesticide solutions:
- “Add buffering solution to correct for improper solution pH.
- Add acid or buffering solution to correct for high alkalinity.
- Add ammonium sulfate (AMS) or 30% nitrogen to correct for hard water. (Generally, add 8.5 to 17.5 lbs dry AMS per 100 gallons water or 1.25-2.5% by volume of liquid fertilizer such as 28%N, 32%N, or 10-24-0).
- Mix pesticides in the field and apply within two hours.
- Filter turbid water or water with high organic compounds.
- Find an alternative water source if mitigation is not feasible.”
For those who’ve asked about using citric acid to adjust pH, University of New Hampshire shares, “Citric Acid (granulated): A food-grade acidifier. Example: 2 oz per 100 gal typically lowers pH from ~8.3 to ~5.4 (always test to confirm).” This guide from Purdue University on Water Quality and Pesticide Performance can also be a helpful resource that goes into more detail: https://www.extension.purdue.edu/extmedia/ppp/ppp-86.pdf.

June 2026 Events
Wheat Variety Tours: Due to drought and frost impacts to wheat throughout the State this spring, the tours have been modified this year. Full details in this article: https://go.unl.edu/bm7f. Tours still occurring for those of you who receive this column include: June 10 for Jefferson County, Knobel Seeds (5 p.m. CT); June 15 for Hitchcock/Red Willow County, Peters Seed Farms (5 p.m. CT); and June 16 for Perkins County, Stumpf International Wheat Research Center (9 a.m. MT). Also a note, because certified wheat seed will be in short supply, it’s recommended that you reserve wheat seed as soon as planting intentions are determined for next year’s cropping season.
Water to Yield Workshop June 11: Farmers and agribusiness professionals are invited to attend a Water to Yield Workshop to be held at the Davenport Community Center in Davenport from 10 a.m.-3 p.m. (registration begins at 9:30 a.m.). The goal is to help producers better manage water and nitrogen resources for enhanced water quality and quantity. Topics include research from UNL’s South Central Ag Lab on irrigation scheduling with limited water, a free app for irrigation scheduling, subsurface drip irrigation, fertigation, in-season nitrogen management, and updates from the Little Blue NRD. Farmers will also share their insights on managing irrigation and in-season nitrogen on a farmer panel. There is no charge to attend, but registration is requested for a meal count. Please RSVP to the Clay County Extension Office at 402-762-3644 or at https://go.unl.edu/dnci.
Summer Stocker/Yearling Tour will be held on June 23 near Broken Bow, offering cattle producers an opportunity to learn from Nebraska operations managing through ongoing drought conditions and explore current research and management strategies for stocker and yearling systems. Registration will be held from 9 to 9:30 a.m. CST at the Custer County Fairgrounds 4-H Building, 44100 Memorial Drive in Broken Bow. Tour participants will visit operations managed by Rodney Lamb at Lamb Farm and Feedyard and Mike Tierney, where discussions will focus on drought management, grazing strategies, forage conditions and decision-making during challenging environmental conditions.
Lunch will be served at noon at the 4-H Building and will include a presentation from UNL beef systems specialists on a new vitamin and mineral balancing tool designed to support producer decision-making and herd performance. The cost to attend is $20, which includes lunch. Registration is due June 19. Participants can register online at go.unl.edu/summerstockertour or by calling 308-268-3105. The tour is sponsored by Merck Animal Health.
Weed Science Field Day will be held on June 24 at UNL’s South Central Ag Lab near Clay Center. The program runs from 9 a.m. to 1 p.m. (Registration at 8:30 a.m.). The field day showcases chemical comparisons for weed control in corn, sorghum, and soybean. I also appreciate how Dr. Amit Jhala is always looking at alternative options for weed control or system’s approaches that include chemicals and cover crops. New this year is an evaluation of the Ecorobotix ARA Precision Sprayer for targeted weed management. Row spacing of 15” vs. 30” with overlapping residual on palmer control and seed production will be shared. Also, hairy vetch termination timing for control of palmer amaranth in sorghum and popcorn will be shared. There is no charge to attend, but registration is requested to: https://agronomy.unl.edu/fieldday/.



Sprinkler Packages and Wind
Grateful for those who paid the ultimate sacrifice for our freedom and to their families left behind. May we never forget that freedom isn’t free! This week sharing excerpts of an article that my colleagues wrote on sprinkler packages in windy conditions. The full article is here: https://go.unl.edu/f90p. “Efficient irrigation relies on minimizing water losses and maximizing the portion of applied water that reaches the crop root zone. …Past UNL research has shown that evaporation losses are often less than 4% of the applied water, though potentially approaching 10% with sprinklers on top of the pipe under high pressures, creating small droplets. In contrast, wind drift can lead to larger losses than evaporation by physically moving water droplets away from the intended target area. This not only reduces irrigation efficiency but can also create uneven water distribution across the field.
…a field evaluation was conducted recently at the UNL’s South Central Agricultural Laboratory (SCAL) near Clay Center, Nebraska. The evaluation was conducted with sustained winds of 10-15 mph and gusts reaching 20-25 mph, conditions that are not uncommon during the irrigation season in Nebraska. The sprinkler industry offers a wide range of sprinkler packages designed to meet different irrigation goals. In this test, three sprinkler configurations from Nelson Irrigation Corporation were tested: 1) Orbitor with blue plate (large droplet design). 2) Sprayhead with tan bubble-wide plate (bubbler-style). 3) Sprayhead with purple plate (fine droplet design).
All these sprinklers were mounted on drops at 9 feet aboveground. Each configuration was assessed based on observed wetted diameter, droplet size and behavior, and overall performance under windy conditions. In addition, a catch-can test was conducted to evaluate how closely the applied water matched the target depth programmed into the system.
The Orbitor sprinkler performed consistently under windy conditions and maintained a stable water distribution pattern. It also produced a relatively large wetted diameter: approximately 40 feet in the direction of the wind. This broader coverage can help reduce application intensity and thus, the risk of generating surface runoff.
Sprayhead with Tan Bubble-Wide Plate: The bubbler configuration produced larger droplets and demonstrated strong resistance to wind drift. The wetted diameter was less than half of Orbitor’s at approximately 18 feet in the direction of the wind. These characteristics make this type of sprinkler particularly effective at delivering water to the intended area when wind is a concern. However, some localized ponding was observed during the test, even at a relatively low application depth of 0.25-inch. This suggests that the higher application intensity associated with a smaller wetted diameter may increase the risk of runoff, particularly in soils with lower infiltration rates or in fields with slope.
Sprayhead with Purple Plate: This configuration produced the smallest droplet size and, under calm conditions, would be expected to create a relatively narrow wetted pattern. However, under the windy conditions during the test, performance was significantly compromised. The fine droplets were highly susceptible to wind drift, to the extent that the boundaries of the wetted area could not be clearly identified in the field. …this type of sprinkler configuration may result in reduced irrigation efficiency and non-uniform water distribution when used in windy environments.
The results of this field test reinforce a key point: sprinkler selection plays a critical role in determining how effectively irrigation water is delivered under windy conditions. In windy conditions, droplet size matters. Sprinklers that produce larger droplets are less likely to be carried away by wind, improving the likelihood that water reaches the soil surface where it is needed. There is a tradeoff between drift and application intensity. While larger droplets reduce drift, they are often associated with smaller wetted diameters and higher application rates, which may increase the risk of ponding or runoff. …Producers are encouraged to evaluate their current systems, consider how they perform under windy conditions, and work with irrigation professionals to select and maintain sprinkler packages that align with your field conditions and water management goals.”


Forage Resources in Drought
Grateful to see some widespread rain this weekend! For fun, there’s a number of tales for predicting rain, including seeing snakes on/crossing the road. Some growers have shared specific numbers with me such as needing to see seven or five snakes on the road. I saw a post on X from the Western Colorado/Wyoming/Panhandle of Nebraska that had a number of replies regarding seeing snakes on the road and many chiming in regarding how many they’d seen last week. There were some who commented about turtles too (of which I also saw one crossing the road last week). And while farmers haven’t mentioned this one to me, it appears that there’s science behind these low-flying, at times kamikaze birds flying so low recently. Air pressure changes before a rain push the insects lower to the ground making the birds to fly lower to eat them. Regardless of what all is truth, it is interesting to me to hear what people observe and to consider how God’s creation knows weather events before we do.
Most of my questions this past week were on the lack of forage with cool-season pastures being so short, mostly in this area of the state. I continue to receive calls from the wildfire and severe drought impacted areas of western Nebraska too. We’re trying to help people with forage resources for grazing and also for drylot space get matched with those who need it at either: https://www.pasturematch.com/ or https://nebraskagrazingexchange.com/.
For people looking for hay and forage in the form of round or square bales, or for those who have bales to sell, a resource is the Nebraska hay and forage hotline at: https://nda.nebraska.gov/promotion/hay.
Received a number of calls about pastures, wheat, and alfalfa going backwards due to drought. We’ll need to watch how the rain changes things, but last week, I was recommending growers have crop insurance look at wheat and then consider getting it grazed or hayed for forage. It still may be a good option considering the need for forage many have.
Much of the alfalfa I’ve been looking at is in the bud stage nearing bloom but is often only 6-8” tall in non-irrigated fields and quit growing. My colleague Ben Beckman and I wrote an article for this week’s CropWatch (https://go.unl.edu/kmcg) where we figured that first cutting alfalfa would need to yield 0.5 ton to pay for the cost of fuel and cutting, raking, and baling. Other universities suggested no cutting of alfalfa if it was less than 10” tall. Quality is maintained in drought-stressed alfalfa, so if you do harvest, waiting for full bloom (vs. 1/10 bloom like we normally recommend) will help drought-stressed crowns recover better. With the recent rains, we’ll have to watch how the alfalfa regrows as it comes out of dormancy.
Grazing alfalfa is an option I’ve recommended to several in this situation instead, if there’s opportunity for fence and water. Waiting till the alfalfa blooms reduces any chance of bloat and waiting till full bloom helps the alfalfa recover better after grazing. Other things to consider when grazing alfalfa are: don’t turn animals out hungry, provide some free choice hay, and avoid turning animals out when there’s dew or recent rain on the leaves (allow the leaves to dry first).
If you can’t graze and can’t justify cutting hay, it’s probably best to just leave it alone. Shredding or haying will give you a cleaner, higher quality hay once your alfalfa does receive enough rain for regrowth. While removing dormant growth may slightly speed regrowth, plants will recover following sufficient rainfall even without cutting. In most cases, it’s not worth the cost and time involved to shred, clip or harvest low-yielding hay.
JenResources 5-10-26
Planting has greatly progressed and/or finished for most in the area. I enjoy seeing the fuzzy green appearance of fields with something growing in them again! The main issues I’m hearing about are drought, crusting, and ground squirrels. We will keep praying for rain! For ground squirrels, I’ve heard the following options for management: plant deeper (2 ¾” vs. they will dig to 2.5”), get a badger!, bait holes and replant. Zinc phosphide is labeled for use for ground squirrels. Nathan Mueller, a former colleague, had shared a pre-plant option (ZP® Rodent Bait AG) for consideration for next year. That product can be applied pre-plant or at planting according to labeled instructions. If you find something that works, please let me know so I can pass it along!
NUE Program: A reminder that the Nitrogen Use Efficiency program deadline to apply is May 15, 2026. The application and instructions can be found at: https://dwee.nebraska.gov/nebraska-nitrogen-use-efficiency-nue-program.
Lawn clippings around plants: Many homeowners apply herbicides (or pay a company to apply them) to their lawns. Herbicides include products that control dandelions, clover, and other weeds in lawns. They are also contained in “weed and feed” products. Most of these herbicides have restrictions that require at least 3-4 mowings of the lawn (so essentially think 3-4 weeks) before lawn clippings can be placed around garden plants. Dying and/or curling leaves around vegetables and flowers may be attributed to people placing lawn mulch around plants too soon.
Mulch and termites: Does using wood mulch around the home attract termites? No as the chipping process kills termites. The most noted study was conducted by Long, et al. in 2001 which compared different types of mulches. They found termites prefer higher nutrient woody materials like cardboard placed under inorganic mulches like pea gravel compared to wood or needle mulch. However, mulches can create a hospitable environment for subterranean termites already established in the soil near the home or building. Termites prefer a moist environment, so soil desiccation (letting the soil dry) near the house foundation is an important barrier. Some keys with applying mulch include: don’t apply more than 2” thick layer of mulch in landscape beds, ensure a 6” gap between the top of the mulch layer and the house siding, and avoid watering mulch or soil close the house foundation walls. Mulch can be applied up to the house foundation to within 12” of the foundation. One may also consult a professional to set up monitoring or barrier control methods for termites around the home or building. The following guide has additional helpful thoughts on termite prevention and control: https://edis.ifas.ufl.edu/publication/IN1277.
Roller Crimping Workshop: Discover how roller crimpers can help manage cover crops without turning the soil — see the equipment in action and learn how to make this low-disturbance method work for your operation at Nebraska Extension’s hands-on workshop on Wednesday, May 20. Free on-site registration begins at 9 a.m.; no pre-registration is required. The program will begin at 9:30 a.m. and conclude at 11:30 a.m. For more information, contact Katja Koehler-Cole at 402-504-1016 or Aaron Nygren at 402-624-8030. ENREEC is located at 1071 County Road G, Ithaca, Nebraska.
Roller crimpers mechanically terminate cover crops while leaving the soil undisturbed, ideally creating a thick mulch that suppresses weeds. They are commonly used by organic and regenerative farmers, as well as others looking to reduce herbicide inputs or improve weed control with cover crops. Growers and ag professionals interested in learning more about this termination strategy are invited to attend this workshop.
Frost Damage to Emerged Corn & Soybean
What to expect from frost on emerged corn and soybean: The cold temps on Friday night resulted in questions on what to expect regarding any potential frost damage to emerged corn and soybeans. For crop damage situations, waiting five to 10 days helps when assessing regrowth potential. The warm conditions of the past weekend following this freeze event can result in a quicker assessment of 3-5 days.
Plant tissues typically do not freeze when the air temperature around them is 32°F. The reason is because solutes are present in the membrane-bound cytoplasm (and also just outside of cell membranes) and they act like a very modest anti-freeze. Thus, plant tissue usually does not freeze until the tissue temperature reaches 30°F to 28°F. Source: Pearce, R.S. 2001. Plant Freezing and Damage. Annals of Botany 87:417-424.
Early season hail and/or freeze damage on corn can cause exposed leaves to be damaged. Fortunately, until V5-V6, the growing point is below the soil and the corn should recover. The damaged leaves will dry and, as regrowth pushes through, the damaged leaves should blow away in the wind. However, wrapping can occur, so fields should be monitored.
In assessing corn plants, dig up and then slice open plants to evaluate the growing point. If the growing point is white or cream in color and firm, the plant should survive. In contrast, if the growing point is brownish-colored and mushy, the plant will not likely survive.
For soybeans, cotyledons are actually somewhat frost tolerant since they are 95% water and fairly thick, so low temperatures are needed for more time to cause damage. However, soybeans that are just emerging with the hypocotyl hook exposed at or just above ground level, are the most at risk for damage. The hypocotyl hook is the area of the stem below the soybean cotyledon. Anything that impacts it will result in seedling death. Watch for plants that have soft, mushy, or pinched hypocotyls. Soybeans can survive the cotyledons being stripped and/or burnt off. Soybean has additional growing points at the axillary buds. If damage occurred below the cotyledons, the plant will die. If above the cotyledons, check for new growth at axillary buds.
What about soybeans where the unifoliate leaves are exposed? Unlike corn, once the cotyledons emerge, all of the potential growing points are above ground so damage could occur. However, the key word in this last statement is growing points, as in more than one. So, even if low temperatures damage the stem tip, there are still two more growing points where the cotyledons attach that the soybean could regrow from. This can set back the plant and cause two stems to form but the plant is still viable if the growing points are still viable.


Picture showing the environmental variability associated with frost damage (Photo by Jenny Brhel).


Pictures taken in 2019 by Jenny Brhel. The soybeans in the left photo had cotyledons just at the soil surface at time of frost. They survived (indicated by thumbs up emoji). The upper right-hand photo shows a seedling with light scarring on the hypocotyl and cotyledons. However, the hypocotyl wasn’t pinched and you can see the plumule between the cotyledons is alive and healthy. The lower right-hand photo shows the hypocotyl was damaged on these seedlings causing pinching. Thus these seedlings didn’t survive (indicated by thumbs down emoji). Soybeans with the hypocotyl hook just at the crack of the soybean surface will have to be watched for how deeply impacted that hypocotyl hook is (if it pinches clear through like in the thumbs down photo).
Frost Damage and Planting
Praising the Lord for some rain! It’s incredible how quickly planting occurs and how much is done. This past week was mostly spent looking at frost damage to wheat, rye, oat, and cover crop forages. Frost damage is worse in fields that are drought and/or moisture stressed and those that have poor stands and/or a poor canopy.
- At 24°F for two hours during jointing in wheat can cause leaf yellowing/burning, death of the growing point, and splitting or bending of the lower stem.
- At 28°F for two hours during boot in wheat can cause the florets to be sterile, heads to be trapped in the boot, damage to the lower stem, leaf discoloration/burning, and odor.
What I’m looking for in the fields: Look for any new growth from the main stem and tillers in the small grains. Split some stems and look to make sure the growing point with the developing head is yellow-green and firm, not brownish and mushy. The good news is that while one tiller may be injured, others on the plant may be ok. The mixture of normal and late tillers can cause uneven maturity though and potential grain yield reduction. Also notice the stem nodes. I have seen even in the better areas of some fields, if the wheat was fairly tall, that some of the lower stem nodes are becoming discolored and splitting. This can cause those tillers to fall over. Those tillers can still provide some nutrient transport to the head, but the lodging can be a pain at harvest and yield reduction will occur. For more information, the following is an article from 2020, but the info. on damage and what to look for is still relevant: https://go.unl.edu/2h7z.
Looking at the week ahead, for those who are planting, be aware of planting windows. We have some cold nights coming after some cold rains this weekend where soil temps have dropped. We say to avoid planting corn 48 hours before a cold snap and soybeans 24 hours before a cold snap. That’s assuming the seed is planted into moisture and begins the germination process. Corn seed needs to absorb 35% of its weight in water and soybean seed needs to absorb 50% of its weight in water in order to complete the initial imbibition (water uptake phase) and be capable of withstanding drops in soil temperatures after that.
Drone Dispatch: Dirk Charlson, our Extension Educator for Digital and Precision Ag, is releasing a newsletter called the Drone Dispatch. Published on the first Tuesday of each month, this newsletter is a product of Nebraska Extension, created to support producers, professionals, and communities using unmanned aerial systems (UAS). Across Nebraska, drones are already transforming agriculture, infrastructure, and emergency response. With that growth comes both opportunity and responsibility and this newsletter is here to help you navigate both. Let’s fly safe, fly smart, and keep Nebraska on the leading edge of drone innovation. If you would like to subscribe to The Nebraska Drone Dispatch, suggest a story or upcoming event, or would like your business or organization featured, please complete the contact from at go.unl.edu/TheNEDroneDispatch.
Forage Resources for Drought and Wildfire: We continue to hear of the need for forage resources between the drought and wildfire impacted areas in Western Nebraska. If you have available pasture and/or cover crop or other forages that you’re willing to custom graze cattle on, please list those resources on both of these places: https://www.pasturematch.com/ and https://nebraskagrazingexchange.com/.
UNL Agronomy/Horticulture Retirements will be celebrated on May 8th with a reception from 2-4 p.m. at the Goodding Learning Center, Plant Sciences Hall at UNL East Campus. Rhae Drijber retires June 30 after 32 years of service. Daren Redfearn retires May 6 after 12 years and Jerry Volesky retires June 30 after 31 years of service to the University of Nebraska–Lincoln. The online guestbooks can be signed at:
https://go.unl.edu/redfearn-retire
https://go.unl.edu/drijber-retire
https://go.unl.edu/volesky-retire
Spring Cold Snaps: How to Handle Your Yard Before and After a Late Frost

Frost can form as ice crystals on plant leaves when temperatures fall below 32-36 degrees Fahrenheit. Photo from Pixabay.com.
By Rachel Anderson, Extension Educator of Horticulture in Lancaster, Cass, Otoe, Seward, and York Counties
Often, the last frosts (32-36°F temperatures) and freezes (28-32°F temperatures and below) of spring pass through our landscapes without much notice or cause for concern. However, such events can affect yards, gardens, trees and lawns in undesirable ways. Follow these tips to minimize damage from cold snaps as winter recedes.
Don’t plant too early.
Check the weather forecast before the average last frost date of the year, which is mid- to late April in southeast Nebraska. As long as nightly low temperatures are set to stay above 32-36°F for the next ten days, you should be safe to plant outside. Exceptions are dormant trees and shrubs as well as many hardened-off cool-season vegetables and annuals, which can be planted earlier in April.
Check that what you plant is proven hardy for your area.
Flowers, grasses, shrubs, trees and other perennials must be adapted to our region’s climate to grow back year after year. Make sure plants are rated for USDA Hardiness Zone 5 or below for eastern Nebraska to ensure survivability through the cold months.
Locate fruit trees and other early blooming woodies in a protected place.
Fruit trees such as cherries, peaches, and plums can fail to produce a crop in years when their flower buds have been damaged by a spring frost or freeze. Help prevent this at planting time by selecting hardy, later-blooming varieties. Locate the plants on north and east facing slopes or sides of structures, which warm up slower in spring, to help delay budbreak until after frost season. This approach can also work for early-blooming, tender-flowered ornamentals like magnolia and forsythia.
Don’t walk or work on frosty lawns.
Avoid walking or driving on lawns with frost on them, as this can damage plant crowns and result in turf browning and dieback. Mowing or fertilizing is not recommended, either.

Walking on a lawn with frost can cause damage that shows up as footprint-shaped dead spots during spring green-up. Photo from Adobe Stock.
Wait to cut back old foliage.
Leaving last year’s stems standing on flowers, grasses, and other herbaceous perennials provides insulation from cold and wind over the winter while serving as habitat for beneficial insects. Don’t remove such material until after the last spring frost (especially with rose and berry canes) or leave at least 9 inches of clearance above plant crowns when cutting back to maintain some protection.

Last year’s stems can be left standing until after the last frost of spring or cut back to 9-12 inches to provide insulation to plant crowns and help prevent frost heaving. Photo from Pixabay.com.
Remove winter mulch and plastic trunk guards.
If you applied extra mulch last fall to protect vulnerable plants over the winter (such as strawberries), make sure to remove it as soon as green growth starts again this spring. Keep in mind that you will need to temporarily reapply such mulch ahead of any forecasted frost or freeze to prevent plant damage.
Plastic trunk collars (typically made of white PVC) put around tree trunks to safeguard against frost cracking over the winter also need to be removed for the growing season. Do this as soon as the risk of frost has passed so that moisture doesn’t get stuck behind the wrap, bringing pest insects or disease.

Remove plastic trunk collars for the growing season as soon as the risk of frost has passed to prevent moisture and pests from collecting behind the wrap. Photo from John Fech.
Signs of Frost Damage and Next Steps
Watch for the following symptoms of plant injury after a late frost or freeze and practice good plant health care to help with recovery. This means keeping plants and trees mulched well past the edge of their leaf canopy with 2-4 inches of shredded wood or leaves, straw, or pine needles (not rubber or rock) and providing deep, infrequent watering so the soil is moist, not soggy or dry. Do not apply fertilizer and refrain from spraying herbicides in the vicinity to minimize the chance of drift while the plant recovers.
Herbaceous Plants
Frost-damaged foliage of non-woody plants first develops a water-soaked appearance, then wilts and collapses, finally turning white, brown or black. For hardy perennials, cut off the foliage after it dies. New shoots will appear on their own in a few weeks. Tender plants such as annuals, tropicals and vegetables, however, typically don’t survive such injury and will need to be replaced.
Frost Heaving
Poorly-rooted perennials can be pushed up out of the ground as the soil freezes and thaws over winter, which can be fatal. Unfortunately, by springtime the damage has been done and you’ll likely have to replace the plants.
Lawns
Damage to lawns from frost alone is rare in Nebraska; however, you may see grass that fails to green up where it was subjected to vehicle or foot traffic over the winter. This often shows up as dead spots in the shape of tire marks or footprints. To help your lawn recover, give it extra time before mowing and cut at a higher height. If the damage turns out to be severe, take steps to mitigate the soil compaction before replacing the grass.
Trees and Shrubs
Trees and shrubs native to the northern Great Plains are well-suited to weather extremes and rarely suffer damage from spring frosts, even if they occur later than usual. However, frost injury to trees and shrubs is possible, especially for less hardy species as well as recently planted woodies that haven’t had time to heel in. In any case, facilitate plant recovery by practicing regular watering and mulching and control pests as needed during the summer to avoid additional stress.

Mulching with shredded hardwood and using a soaker hose to provide deep, infrequent watering are standard care practices that can help trees and shrubs recover from frost damage. Photo from John Fech.
LEAVES: Frost-damaged leaves that have already emerged from their buds at first appear water-soaked and may be darker green in color even as they dry up, sometimes turning black or brown. Frost-damaged leaves that haven’t emerged yet will exhibit browning or leaf tatter later when they eventually grow, which should not be mistaken for insect or disease problems. Do not prune out damaged leaves—allow the plant a few weeks to re-leaf on its own.

Frost damage exhibited by the blackened, curled new shoots of a young walnut tree. Photo from Adobe Stock.
FLOWERS: Frost-damaged flower buds turn dark brown, shriveled or dry, while open flowers go brown, limp and mushy. Woody plants do not regrow new flower buds if the original set is damaged. In the case of fruit trees, such damage provides an entry point for serious diseases like fire blight, so plan to treat accordingly. Otherwise, a loss of flower buds has little negative impact on overall tree and shrub health and doesn’t require intervention.
TWIGS: Frost-damaged twigs and branch tips typically turn darker in color and develop a sunken and wrinkly, ribbed surface. Woody stems exhibiting such symptoms will likely not recover and should be pruned out in late spring after the plant has had time to recoup. To check the extent of the damage, gently scratch a small area of bark away with your fingernail. If you see green tissue, the branch is alive and should be left on. If underneath the bark is brown, the branch is dead and should be pruned back to the closest live bud or side branch.
Frost Cracking on Tree Trunks
Frost cracks that separate a trunk’s bark from its inner wood can appear over winter on some trees. While healthy specimens can close cracks in time with callus tissue, such a wound on a tree already struggling may mean it will never thrive. To increase chances of recovery, follow good care practices related to watering and mulching and do not apply paint or wound dressing. Remove any ragged edges or loose hanging bark from the crack with a clean cut using a sharp, sterilized knife.

Damage from frost cracking, shown here as a split along the trunk, can severely impair a tree’s long-term health. Photo from John Fech.
Evergreens
Symptoms of frost damage to evergreens can appear right away as dying and curling shoot tips, sometimes occurring only on the windward side of the plant. Or it can show up later as wilting and browning of new growth that is often mistaken for herbicide damage. Streaks of white sap on branches can also indicate freeze injury. As correction, prune any curled tips and dead twigs back to a live bud or side branch and provide regular watering and mulch.

Browning and curling of new shoot tips can be a sign of frost injury on evergreens, as with this spruce tree. Photo from Minnesota Department of Natural Resources, Bugwood.org.
2026 Planting Considerations and Soy Seed Treatments
The warmer air, wind, and warming trend of soil temperatures have allowed for planting to begin for some. Last week I mentioned watching the soil moisture where seed will be placed as pre-irrigation may be necessary with the lack of rain most of us received. The winds continue to remove surface moisture and each field varies in soil moisture at seeding depth.
The decisions we make at planting impact the entire season. Aiming for fit soil conditions that are proper and even soil moisture where the seed is placed, warming trend of soil temperatures, and even planting depth are important. We saw how uneven emergence was last year with uneven moisture at the seeding depth. I recommend putting corn and soybeans in the ground at 2” (our research recommends 1.75” for soybean). This allows for buffered soil moisture and temperature conditions when planting. Bob Nielsen, emeritus professor at Purdue said corn can be seeded 2.5-3” deep if that’s where uniform soil moisture is located in order to achieve uniform germination and emergence, particularly for non-irrigated fields. Don’t plant soybean below 2.5”.
Since 2004, we’ve shared the importance of planting soybean early (mid- to late-April or first week of May) to increase yields. We often hear a preference of planting corn and soybeans in as close to 50°F or greater soil temperatures as possible. Below 50°F, I prefer to see corn planted on a warming trend of 5-7 days. The consideration is for soil temps in the mid-40’s on a warming trend with no chance of a cold snap (cold rain/snow) within 8-24 hours for soybean and 48 hours for corn. The time-frame is due to the imbibition (critical water uptake) time-frame for corn and soybean. Soil temps for your field can be monitored by using a thermometer or checking out CropWatch soil temps at: https://cropwatch.unl.edu/soiltemperature.
Soybean Seed Treatments: From 2023-2025, growers have conducted 11 site-years of Nebraska on-farm research soybean seed treatment studies (Table 1). The studies were conducted in Seward, York, Polk, Hamilton, and Clay Counties with planting dates ranging from April 23 to May 30. The goals for the growers were to evaluate the economics and yield resulting from the seed treatments. Some of these growers also desire to move away from traditional fungicide/insecticide seed treatments due to potential impacts to pollinators, soil health, and human health. Some were also interested in any increased plant health due to early seed and microbial associations with a biological seed treatment that may have influenced a healthier rhizosphere microbiome (Berendsen, 2012). Only yield and any presence of disease was assessed.
Results: The results of Table 1 show that in only 1 site-year (Seward-1 2025) the yield of the full company seed treatment out-yield the biological or untreated seed with which it was compared. The cost of the treatments varied by grower due to the products applied to the seed and the seeding rate. Planting dates varied on the year and location, with the locations in 2024 receiving more spring rainfall with later soybean planting. The locations in 2025 were generally planted into warm, dry soil conditions. Soybean disease was not observed as a problem in any of these on-farm research fields even though several of the locations in 2023 and 2024 had a history of white mold. Try this yourself! This is easy to do by splitting a planter or skipping planter passes with treated vs. untreated seed. Please contact me if you’re interested in trying this for on-farm research! Full article: https://go.unl.edu/uyp7.
Treatments Used:
- Full company seed treatment (Cost: $13.72-29.00/ac)
- Untreated Seed (No Cost)
- Biological seed treatment in all the locations other than the two listed in the next bullet point (created and treated by one of the growers): blend of 2 oz PhycoTerra® ST, 1 oz Heads Up®, 1 oz N-Gage Ultra ST, 0.75 oz Bio ST VPH in 100 gal solution. In a second tank, 2 oz of Exceed Soybean inoculant was used per 100 gal only for this treatment. Cost: $9.00-$9.20/ac
- Two other biologicals used included a seed treatment by Elevate Ag (Clay 2025) (Cost: $34/ac) and a home-made compost extract seed treatment (Lancaster 2025) ($0.63/ac).
Table 1. Eleven Site-Years of Nebraska On-Farm Research Seed Treatment Yield Data
| Location, Year | Number of Reps | Planting Date | Full Company | Biological | Untreated | Full Company Cost ($/ac) | Biological Cost ($/ac) |
|---|---|---|---|---|---|---|---|
| Seward, 2023 | 6 | May 2 | 62A | 61A | no data | $28.00 | $9.20 |
| York, 2023 | 6 | May 10 | 66A | 67A | no data | $28.00 | $9.20 |
| Hamiliton, 2023 | 6 | May 22 | 69A | 68A | no data | $16.63 | $9.20 |
| Seward 1, 2024 | 4 | May 10 | 71A | 69A | 70A | $27.00 | $9.00 |
| Seward 2, 2024 | 6 | May 30 | no data | 70A | 70A | no data | $9.00 |
| Polk, 2024 | 4 | May 27 | 77A | 75A | no data | $17.10 | $9.20 |
| Clay, 2025 | 6 | May 5 | 71A | 68B | 70A | $18.50 | $32.14 |
| Seward 1, 2025 | 4 | April 29 | 76A | 73B | no data | $29.00 | $9.00 |
| Seward 2, 2025 | 3 | April 28 | 76A | no data | 75A | $13.72 | no data |
| York, 2025 | 3 | April 28 | 77A | no data | 76A | $13.72 | no data |
| Lancaster, 2025 | 4 | April 23 | 63A | 63A | 63A | $18.51 | $0.63 |
Table 1 Note: Same letters are not statistically different at 90% confidence level. Analyzed by each individual location.
Berendsen, Roeland L., Pieterse, Corne´M.J., and Bakker, Peter A.H.M. August 2012. The rhizosphere microbiome and plant health. Trends in Plant Science Vol. 17, No. 8, pg. 478-486.
