Category Archives: Crop Updates
2025 Yields Part 2
York County Corn Grower Banquet will be held Thursday, November 20th at Chances ‘R in York. Social at 6:30 p.m. with meal at 7 p.m. Tickets are $20 and can be reserved at the Extension Office at (402) 362-5508 or contacting a York Co. Corn Grower Director.
In last week’s column, we walked through the growing season providing perspectives of what may have caused yield loss. I mentioned that while people are looking for solutions, increased nitrogen rates, more fungicide applications, and tillage are not the answers for higher yields. In conversations, those were the “solutions” people were trying to provide. Several asked me for any patterns from observations and conversations I’ve had. That’s what I will attempt to share in this column. There’s a number of field situations where only 150 lb N and one fungicide application resulted in above-average yields. In those situations, correlations included balanced overall nutrition where nitrogen was reduced, TIMELY fungicide application, and not over-irrigating.
I realize quite a bit of tillage has occurred. Gently, please remember the wind events we receive each spring. I encourage you if you have tilled, to consider covering the ground with cereal rye. March 2025 was the windiest March on record in Nebraska and I remember several accidents due to poor visibility. I realize this is a hard topic. Tillage will not control southern rust as that fungus doesn’t overwinter in residue. It will help with the tar spot fungus, but there are other management considerations including hybrid and not over-applying nitrogen and irrigation.
Reduced nitrogen: It was interesting walking our on-farm research studies where we had nitrogen ramps this past year and observe the correlation between less southern rust in the lower nitrogen rates and more southern rust in the higher nitrogen rates. Dr. Bob Gunzenhauser observed the same thing this past year and posted a photo on X showing two hybrids and the variation of southern rust compared to nitrogen rates.

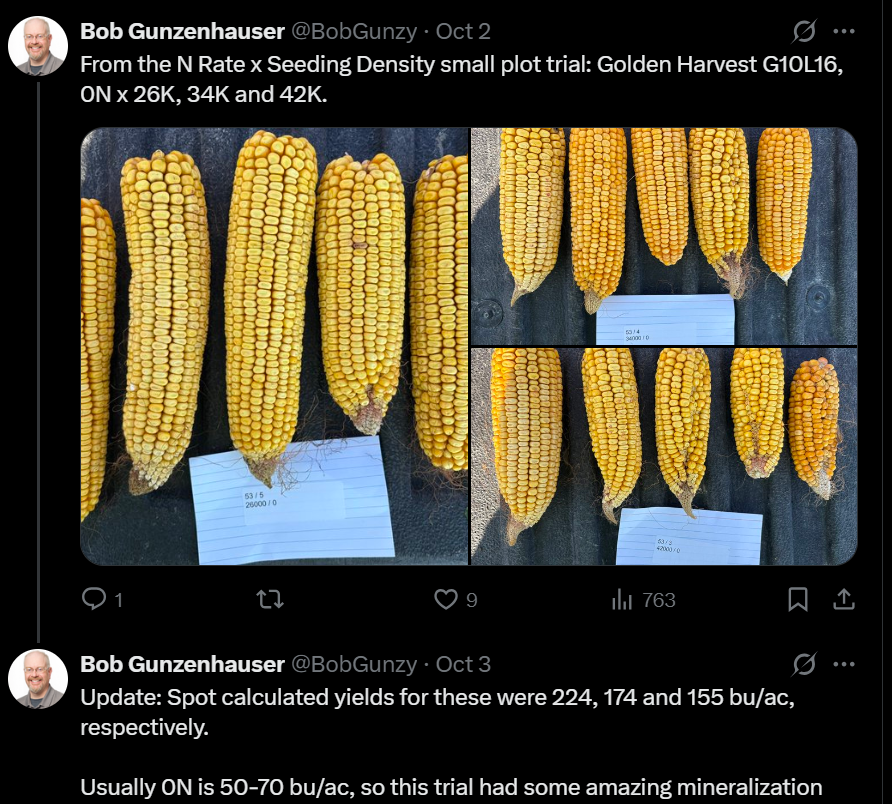
Timely fungicide application: There were people with better yields (250+) who applied only one fungicide while others applied two. I didn’t hear too much from those who applied three to five applications. Some applied two fungicide timings of two different generic products and did just as well as the higher priced products with one application. The key was not applying the fungicide too early so there was some later season residual. Some applied a second fungicide around late dough to early dent stage this year and that seemed to work for hybrids that were more prone to southern rust. The keys were knowing the hybrid, not having excess nitrogen, and proper irrigation management.
Irrigation Management was perhaps the thing that was most frustrating for growers. We started the season off so dry after several years of being dry. So, it’s easy to be in the mindset of irrigating. Anytime non-irrigated fields yield the same or more than irrigated fields, it’s often due to over-irrigation. Irrigation events often occurred before rain events this year. This year’s relative humidity was also very high, thus, the crop didn’t use as much water. Because of that, we were keeping some soils too wet by irrigating which, both Dr. Tom Hoegemeyer and I believe, added to the crown rot and stalk rot issues we experienced. Dr. Hoegemeyer also wrote a recent article reflecting on “60 Harvests – Changes I Have Seen — Nebraska Soil Health Coalition“.
Final thoughts, there’s not one answer but a combination of factors that impacted fields this year. There’s also a lot of farmers hurting with the combination of low yields, low commodity prices and high input costs. Hundreds of thousands of dollars were lost per field in several situations. For those involved and/or interacting with the farming community, please reach out to each other and check in with each other. Please know you’re not alone and there’s always help and hope if you’re struggling. 988 is Nebraska’s suicide and crisis lifeline. There’s also a number of people who have announced retirements or will be announcing them. It’s important to be supportive. Conversations I’ve had with growers this year include: retiring to avoid taking on additional debt, wanting to scale back to reduce debt and stress, and retiring due to health reasons-both physically and anxiety/stress. It’s important for those retiring to retire “to something” by staying active in some way.



2025 Yields
This harvest was a difficult one, plagued with breakdowns, slow-going in storm-damaged corn, and disappointing yields. While there were a few reports of decent yields, the overwhelming majority of farmers and seed dealers in the area have been disappointed. Honestly, I was worn down and needed a mental break before I could address this in writing. I asked Dr. Roger Elmore, Dr. Tom Hoegemeyer, Dr. Bob Nielsen, and Dr. Eric Hunt if my reasoning was on track and for their additional thoughts and am grateful to them.
Major Point: People are looking for solutions, but increased nitrogen rates, more fungicide applications, and tillage are not the answers. What went right? Balanced nutrient management with reduced nitrogen inputs, TIMELY fungicide application, and proper irrigation management are future keys.
We began the season with dry planting conditions. I was arguing we were potentially drier than Spring 2023 with the observations about rye and pastures not growing. Crops went into the ground quickly without cold snaps. Several farmers were completely done planting in April. Irrigation began early to get moisture into seedbeds and to activate herbicide. Plant stands and emergence were uneven, evidenced again at harvest with varying ear sizes and plants with ears that didn’t pollinate. I think that impacted us more than we realized. The Memorial Day weekend rains saved us.
A relief was that rain kept falling in spite of it varying greatly in timing and amounts. Some experienced higher non-irrigated yields in corn and soybeans compared to irrigated fields. That nearly always is due to too much irrigation and timing of those irrigations, often occurring right before a significant rain event.
We had a few wind/hailstorms in July and the fairly widespread Aug. 8-10 wind/hail event, which York Co. escaped. Much of the year we received lower than average solar radiation (which includes photosynthetically active radiation or PAR). There were several periods of cloudy/hazy/smoky days. Research utilizing shade cloth revealed 25-30% potential yield loss with shading occurring from R2-R6 stages in corn. As Dr. Roger Elmore pointed out, the hybrid maize model was predicting average yields at the end of the growing season in spite of the low PAR, which would suggest biotic (living) factors being the greater issue. Photosynthetic stress on plants can also include southern rust impacts on leaf tissue and stalk rots. I’m wondering if irrigation prior to heavy rain events exacerbated the fusarium crown rot/gibberella stalk rot we saw? Dr. Tom Hoegemeyer wondered the same thing. “Photosynthetic stress and stalk rot go together like beans and weenies. Each one can cause the other. We MAY have had some early infection with Fusarium/gib due to saturated soils/etc. As you know, high N rates, lower K available and a dozen other stress sources make it worse.”
High night-time temperatures burn sugars that should go into ears to fill kernels. I mentioned my concern about this throughout July and August. By mid to late August, ears began pre-maturely drooping, cutting off the food supply to kernels. Looking at kernels in numerous fields at harvest time, they appeared shriveled/pinched at the base. Dr.’s Tom Hoegemeyer, Roger Elmore, and Bob Nielsen all attribute that to stress occurring before black layer in which the kernels prematurely died before completing the normal black layer process. I feel the greatest contributors of this were the high night-time temperatures and the stresses of southern rust and stalk rots. Dr. Eric Hunt also mentioned the high humidity, particularly in York County due to the sheer amount of irrigation which may have led to increased disease pressure including stalk rots.
Dr. Bob Nielsen: “Your description of the kernels makes me think that kernel development was prematurely halted. Although, honestly, severe reductions in photosynthetic leaf tissue prior to BL (black layer) due to southern rust etc. or early onset of severe stalk rots would also prematurely shut down kernel development. And, of course,…(large) ears with excellent kernel set create a huge demand for photosynthate during grain fill, which exacerbates the negative effects of severe loss of photosynthetic leaf tissue and predisposes the stalk and root tissue to rapid fungal rot infection and development.”
Dr. Tom Hoegemeyer summed it up: “I think we had lots of issues that caused PS (photosynthetic) stress, some of which impacted our irrigated acres worse than our dryland acres. (My home dryland area had lots of 200 to 220 bpa corn and 65 to 70 bpa soybeans. After a dry spring, we had more rain than we’ve had for years). Irrigated corn in the area often wasn’t as good as the dryland, even with more N applied. The more stressors (hot nights, light limitations, too high N for the amt of light/PS–exacerbating disease issues, multiple leaf diseases combined with high humidity, continuous corn, etc.) the bigger the yield loss. And, in some instances, I think adding water to these fields hurt more than it helped.”




Sources:

York: -21 MJ/m2
Grand Island: -9.7 MJ/m2
Lincoln: -25.2 MJ/m2
Falls City: -28.3 MJ/m2
Norfolk: -10.7 MJ/m2
Wayne: +19.7 MJ/m2
West Point: -1.3 MJ/m2″



Corn Dry-Down
With harvest ramping up this week, wishing you safety and a good harvest! Make sure to think fire safety and also take care of yourselves emotionally, mentally, and physically. Taking breaks will be important, especially when harvesting this storm-damaged corn, but also because of the stress many are facing.
Watch crop harvest moistures: I mentioned the importance of watching soybean moisture last week and checking soybeans when there’s still some leaves remaining on plants in spite of green stems. We updated this CropWatch article last week https://go.unl.edu/dzb4 and added a table to better visualize what’s being given up in gross by selling soybeans above and under 13% moisture.

Corn is also drying faster than what one may realize, especially after this weekend’s hot temperatures and because of the amount of late-season disease. I’ve been asked what to expect for dry-down for those using storm-damaged corn for high-moisture corn and/or silage. Upon reaching physiological maturity (black layer), most corn hybrids are around 30% moisture. The average corn dry-down for hybrids reaching maturity in late August is around 0.8 percentage point per day and 0.4 percentage point per day for hybrids reaching maturity in mid-September. High heat and wind, such as we experienced last year in September 2024, can lead to well over 1.0 percentage point per day in dry down.

Accelerated senescence and grain drying of corn can also occur with the hail damage and lodging in addition to high amount of late-season southern rust and tar spot this year. One can easily observe hybrid differences in how they’ve reacted to southern rust. Sometimes these pressures lead to premature death of plants including the premature formation of black layer. When that happens, grain drying occurred earlier than anticipated with a higher dry-down rate per day than if the grain had matured normally.
Corn hybrid characteristics can also impact kernel moisture. Hybrids that have thinner or permeable pericarp layers (outermost layer of the corn kernel), are more subject to moisture loss and faster drying rates in the field. I’m unsure how one knows this characteristic though. Other characteristics resulting in quicker dry-down include: lower husk leaf number and thinner husk leaf, how soon the husk leaf tissue turns brown, how well the husk covers the ear (less coverage allows for faster drying), how tight the husk is (as looser husks will increase moisture loss), and how fast the ear turns down vs. remaining upright.
My concern, particularly in the non-irrigated fields, storm damaged stripped up corn, and corn heavily impacted by southern rust in the area I serve, is that corn is actually dryer than one may realize. Apparently this concern also exists in Indiana as Dr. Bob Nielsen, Professor Emeritus from Purdue University, released an article last week on avoiding selling too dry of corn to the elevator: https://www.agry.purdue.edu/ext/corn/news/timeless/WaterShrink.html.
Alfalfa and Wheat Expo: Nebraska Extension is hosting the 2025 Southeast Nebraska Alfalfa & Wheat Expo on Friday, Sept. 19, from 8:30 a.m. to 3:30 p.m. at the Saline County Fairgrounds Office (1600 Tuxedo Park Rd., Crete). This one-day event is designed for wheat and alfalfa producers to gain practical insights, connect with experts, and sharpen their management strategies. Topics include: Equipment considerations for wheat production; Wheat disease updates and management strategies; Insights on achieving high-yield wheat in southeast Nebraska; Alfalfa irrigation, insects, weeds, and crop health management; Pesticide safety updates; Sponsor exhibits, networking, and raffle prizes. Come for the whole day or stop in for the sessions that fit your schedule. Register online at https://go.unl.edu/2025-alfalfa-wheat-expo or call the Saline County Extension office at 402-821-2151.



Harvest Soybean Moisture
What a beautiful September we’ve had thus far! Seed corn harvest has begun and non-irrigated corn is rapidly turning. Soybean harvest will most likely occur by this weekend the way early-season beans are turning.
One note, I’ve noticed droopy ears for several weeks now. It can be due to various environmental X genetic factors such as the high night-time temps coupled with cloudy weather both impacting photosynthesis with specific genetics. I also often observe ear droop to be associated with Fusarium crown rot. Estimated yield losses can be from 12-40% per ear (depending on if the shank pinching occurred during late or early dent stage, respectively). Also, scout fields for shank integrity and seek to prioritize harvest of fields with any increased risk of ear drop.
As we approach soybean harvest, perhaps a frustrating, yet important topic, is seeking to harvest soybeans as close to 13% (the elevator standard) as possible. The frustration is it seems to be such an art and luck in order to do so. For the last several years, it seems that the beans are often drier than what one realizes in spite of green stems and sometimes even leaves remaining on the beans. Test the beans for moisture earlier than you think they may be ready. There’s years like last year when I didn’t even bring this topic up with the hot, dry winds. Hopefully we don’t have a repeat of that.
To avoid beginning harvest when the soybeans are too dry, consider beginning harvest at 14% moisture making combine adjustments and operating at slower speeds as necessary. While there is a dock of around 2.5% for the first 2 points delivering wet beans (the percentage differs by Coops), delivering soybeans much below 13% moisture reduces profits because there’s fewer bushels to sell (load weight divided by 60 lbs/bu assuming 13% moisture). Selling soybeans at 8% moisture, you’re losing about 5.43% yield; at 9% moisture, it’s 4.4%; at 10% moisture, 3.3%; at 11% moisture, 2.25%; and at 12% moisture, it’s 1.14% yield loss. That doesn’t take into account additional risk for shatter losses during harvest. Only 4-5 beans on the ground can add up to a bushel/acre loss due to shatter. The following are profit examples for a field that’s yielding 75 bu/ac:
Example 1. If the grower was to sell beans at 13.8% moisture, he/she could be docked 2.5% of the selling price of $9.60/bu, reducing the actual price to $9.36 per bushel. Total income per acre would be: 75 bu/ac yield x $9.36/bu = $702 per acre gross
Example 2. If the soybeans were harvested at 9% moisture, there would be 3.3 fewer bushels per acre to sell (4.4% of 75 bu/ac yield due to water loss): 75 bu/ac – 3.3 bu/ac =71.7 bu/ac yield x $9.60 = $688.32 per acre gross. In this example it’s better to take a dockage for selling beans at 13.8% moisture than sell them at 9%. The difference is a positive gain of $13.68 per acre or around $1846.80 on a 135 acre field.
Example 3. If the soybeans were harvested at 12% moisture, there would be 0.86 fewer bushels per acre to sell (1.14% of 75 bu/ac due to water loss): 75 bu/ac – 0.86 = 74.14 bu/ac yield X $9.60 = $711.74 per acre gross. If you can’t hit 13%, it’s still pretty profitable to sell them for 12% moisture compared to the other examples.
One more note, we tend to see around 1 day delay in harvest for every 0.1 difference in maturity group. Here’s wishing you a safe and profitable harvest!




Various Resources
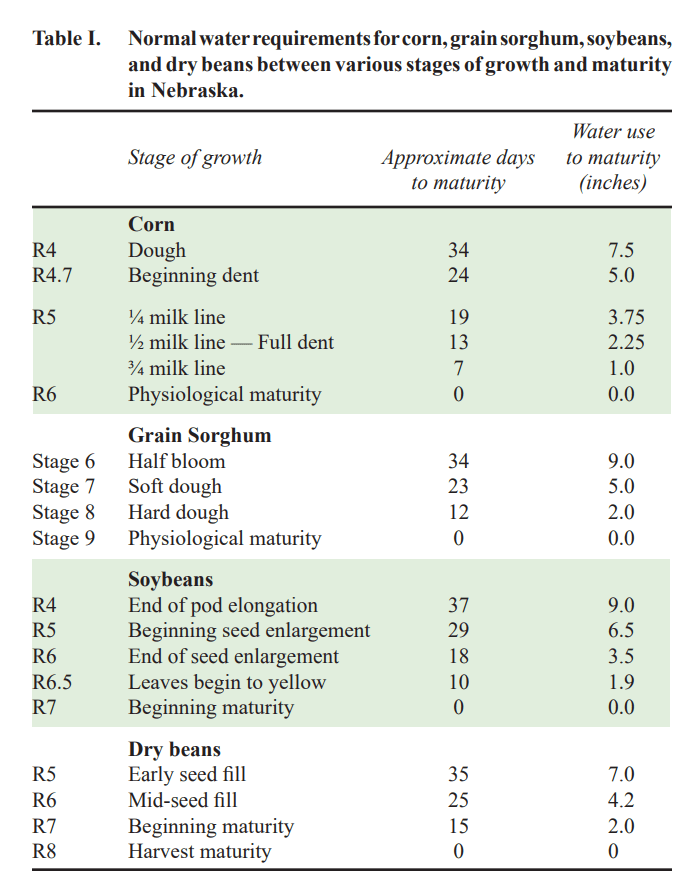
Water use till maturity for Corn: Beginning Dent 5”, ¼ milk 3.75”; ½ milk 2.25”; ¾ milk 1”.
Water use till maturity for Beans: Beg. Seed 6.5”; Full Seed 3.5”; Leaves beg yellow 1.9”.
The Nebraska State Fair is occurring and this past week/weekend was a full one for the 4-H side with FFA projects to be showcased next weekend! Extra special to me are the 4-H identification contests where youth compete to show their knowledge. This year, the York County 4-H youth received Champion Intermediate Horticulture ID team, Champion Intermediate Weed ID team, and Reserve Champion Senior Weed ID Team! So proud of all the youth who competed this year and in the past!
Southern rust of corn was the most frequent question I received last week. The recommendation from research shows there’s no additional benefit to spraying when the corn is two weeks prior to full dent. https://cropprotectionnetwork.org/publications/an-overview-of-southern-rust. Corn is also rapidly progressing in maturity due to the high night-time temperatures we’ve had. This results in corn plants burning sugars at night instead of filling kernels as deeply which can impact yields.

Leaf spot on lilacs: have received a number of calls about lilacs that appear to be “dying”. The culprit is pseudocercospora leaf spot, which is caused by a fungus. The leaves will brown and fall off the lilacs and you will most likely see new growth occurring. Stems are still pliable and buds are still green. Nothing to do other than to rake up the leaves that fall as the fungus can survive in the leaves for two years. More detailed information: https://extension.missouri.edu/media/wysiwyg/Extensiondata/Pro/PlantDiagnosticClinic/Docs/leaf-spot-on-lilac.pdf.
An additional note, fungal leaf spots are found on a number of trees and shrubs right now due to the humid conditions with heavy dew. You may observe impacted leaves fall off early with new growth beginning on the trees/shrubs. Again, nothing to worry about or do for now.
Fall Armyworms have been on the rise over the past few weeks in pastures, alfalfa, lawns, and some crop fields. It will be important to keep watching newly seeded small grain, alfalfa, and cover crop fields this fall. Caterpillars ¾ inch or longer are close to maturity and can be harder to control with an insecticide. There’s a number of conventional and organic insecticides that can be used. Products with 0 day grazing restrictions in pastures include Warrior II, Mustang Max, Beseige, and Prevathon. While it’s not updated, a Q/A for consideration can be found here: https://cropwatch.unl.edu/2021/fall-armyworms-pastures-alfalfa-small-grains-cover-crops-and-lawns/.
Resources: Wanted to share a number of resources. First, a reminder if you have a verbal crop lease you wish to terminate, that must be done by September 1st. More specific information here: https://cap.unl.edu/news/terminating-verbal-farmland-lease-nebraska/. We recommend transitioning verbal leases to written ones and a great resource to help you do that is Ag Lease101 https://aglease101.org/doclib/ where they have various free templates to help.
There have been a number of recent webinars regarding how the Big Beautiful Bill impacts ag. In case you missed them, here’s some recorded webinars:
- Impacts of the Big Beautiful Bill to Ag: https://cap.unl.edu/more-than-one-big-beautiful-impact-ag/
- Changes to Ag Taxes: https://cap.unl.edu/lets-talk-taxes-webinar/
York County Corn Grower Plot Tour and Shrimp Boil will be held September 2, 2025 from 5:30-7:30 p.m. just north of 1603 Road F, Bradshaw, NE. Seed reps will showcase their hybrids, we’ll hear an update from Nebraska Corn, and enjoy a delicious shrimp boil meal (shrimp, sausage, corn, and potatoes). Guess the highest yield without going over and win a $50 gas card. Must be present at the winter Corn Grower Banquet to win. All are welcome! Members are asked to bring a friend and potential member.





Cloudy Days and Corn Tip-Back
Received a number of questions about tip-back in corn and also how the cloudy/smoky days could impact yields. First wanted to mention the difference between pollination concerns vs. tip-back concerns.
Pollination concerns are when the green silk remains attached to the ovule that should have formed a kernel if it had pollinated. Gently husking back an ear and shaking it will reveal where pollination occurred on the ear where the brown silks detach. Green silks remain attached where pollination did not occur. Tip back is when the ovule pollinated but the kernel later aborted (looks shrunken/shriveled) due to some reason, often some type of stress.
This week’s UNL CropWatch shared yield potential for different places in the Midwest using weather modeling and comparing it to a 30-year average. The data clearly shows low solar radiation (which includes photosynthetically active radiation (PAR)), meaning photosynthesis is reduced. In spite of that, the article is suggesting for non-irrigated corn, nearly 100% of above-average yields for the Beatrice area, and nearly 75% chance of above-average yields near the Clay Center and Mead areas. For irrigated corn, it’s estimating 50% near-average yields for Clay Center, 75% near-average yields for Beatrice, and 50% above-average yields for Mead. These estimations are just based on weather and don’t account for stand losses, disease, insects, etc. The full article is at: https://go.unl.edu/tkz6.

Dr. Roger Elmore and Dr. Tom Hoegemeyer wrote a CropWatch article several years ago about the impacts of cloudy days on different stages of corn development. The full article can be found at: https://go.unl.edu/sv06 and the following are excerpts from it.
“Early et al., 1967,…shaded plants around the “reproductive phase” for 21 days as well as during the “vegetative stage” for 54 days and the “maturation phases” for 63 days. Shading during reproductive stages reduced plant yields the most, but 30% shading during the maturation stages ― what we consider the seed set and grain-fill periods (R2-R6) ― not only reduced yield per plant 25% to 30% but also reduced kernels per plant and the amount of protein per plant.
Researchers in a new study shaded plants from silking to maturity (R1-R6) (Yang et al., 2019). They also found reductions in yield and biomass with more shading resulting in more losses. Shading reduced yields more with higher plant populations than with lower populations.”
If you’re seeing tip-back concerns, it could be due to the consecutive days of cloudy skies reducing photosynthesis. But it’s also important to understand the hybrids in your fields. Hybrids are very specific in how they handle stress such as heat, drought, and cloudy weather during and shortly after pollination. Some hybrids have long ears, and in counting kernels, 30-40 kernels per row with a 1-2” tip-back may be “normal”, especially if there are a larger number of rows around for a decent total kernel number per ear. Plant populations also can play a roll in tip-back. So, not all tip-back is a concern. Knowing the hybrid and counting total kernels per ear can tell a bigger story. We will have to wait and see how the remainder of the grain fill season progresses to better understand any potential yield impacts from the cloudy days.



Crop Update 7/21/25
Crop Update: Pollination is occurring and with the news on social media, received a number of calls regarding corn tassels this week. Last year, I had seen some “tassel wrapping” in a few hybrids but hadn’t said anything as I didn’t receive calls on it. This year it’s far more widespread throughout the country. What is meant by tassel wrapping is the last few leaves are tightly wrapped around the emerging tassel so that only a small portion of the top is pollinating and showing. As I’ve watched these tassels the past two years, they try hard to emerge from the leaves. Some hybrids are able to push out of the leaves better. Some will push out through the side of the leaves. The tassels then look strange with what parts are able to shed pollen once they emerge from the wrapped leaves.
In some of the hybrids, there’s enough pollen being shed from refuge plants and the tassel tops, that they should still pollinate ok. There’s a few with less pollen shed, yet there’s also corn pollinating in nearby fields. Hopefully there won’t be pollination issues. It will be important to watch your specific fields for pollination.



No one likes to see these things happen. There’s been a number of things shared as to what’s going on. Some are calling this environmental. I’ve heard a number of people talking about either generic dicamba or status applications to corn. I’ve been in the fields looking at this for 10 days and would encourage you to walk into the signed plots of the different companies and local County Corn Grower plots. If this was an environment or dicamba issue alone, entire counties of acres would be impacted because of being in a similar environment and because of how much dicamba is used in post-herbicide applications. This unfortunately appears to be genetic as I’m only seeing it in specific genetics. When across companies, they’re genetically related.
Southern rust was also found in Nebraska last week and in our area, it was found in Butler and Polk counties even though Butler isn’t lit up on the map. It’s at low incidence and not a level to spray yet. I’m still saying to wait till at least milk if you’re still planning on a fungicide app to reduce potential pathogen resistance and for best economic thresholds: https://corn.ipmpipe.org/southerncornrust/.
Summer Grazing Field Day: Alex Daake in the Utica area is hosting a field day on Tuesday, August 5th from 4-6 p.m. at 4136 Superior Rd, Utica. He took a pivot and divided it into quarters. One quarter is soybean, corn, grazed mix for 1 year, grazed mix for 2 years. Cereal rye is also taken for seed after the grazing. He rotates the quarters each year. The field day will primarily focus on how Alex is grazing covers, the cover crop mixes used, and how he feels it’s improving the soil. Come learn about what he’s doing, why, and the economics. On the quarter with corn was 2 years of previous grazing. He has an on-farm research nitrogen study on that ranging from 0-150 lb N/ac. He is also using satellite imagery from Sentinel Nutrient Mangement to determine which plots, if any need additional nitrogen. It will be a great afternoon of learning and sharing! Please RSVP to me at jrees2@unl.edu or 402-440-4739 if you’re interested in attending. Cookies and drinks will be provided.
South Central Ag Lab Field Day will be held Thursday, August 7th near Clay Center from 8:45 a.m.-3:10 p.m. (Reg. 8:30 a.m.). The day is designed so you can pick and choose which topics you’d like to attend. More info. at: https://enreec.unl.edu/south-central-agricultural-laboratory-field-day/.
Tar Spot of Early Corn Update
Received several calls about tar spot yesterday and today. As of right now, it’s been confirmed by UNL at LOW incidence (1-2 lesions per leaf) in Saunders, Pierce, and Clay (on 6/16/25), Polk, and Seward Counties (on 6/17/25). I really appreciate Craig Anderson and Mike Byers bringing me leaf samples to confirm. I also appreciate those who were calling to hear of any confirmations from leaf samples.



There’s a lot of fear surrounding this disease, and still some unknowns. We haven’t seen tar spot in Nebraska this early. It would be helpful if consultants/agronomists would confirm samples to Dr. Tamra Jackson-Ziems, the UNL Plant and Pest Diagnostic Lab (tar spot testing is free), or a local Extension Educator so that we have the most accurate information to provide. Tamra will update the tar spot map at: https://cropprotectionnetwork.org/maps/tar-spot-of-corn.
We are NOT recommending fungicide application in fields where tar spot is confirmed in these early vegetative stages.
Reasons for not applying fungicide now:
1-Economics: Corn economics are already a struggle. When tar spot appeared in the early vegetative stages, research from Dr. Darcy Telenko’s lab at Purdue University showed it wasn’t economical to apply at V6-V7 as it didn’t suppress disease enough. It was economical when the corn reached tassel stage or beyond. I show the research data below and you will also see the chart on the website link I shared above. In this post, Dr. Telenko shares 7 years of experience dealing with tar spot when it occurs early in the season and how to make fungicide decisions.
*Some Quick Tips & Tools for Preparing for Tar Spot in Corn-Dr. Darcy Telenko
*Tar Spot: What to look for in corn and making an informed fungicide application-Dr. Darcy Telenko
*Crop Disease Forecasting Tool for Tar Spot
2-Applications: Dr. Tamra Jackson-Ziems shared that some products say “no more than 2 applications per year”. Using those active ingredients now would mean you could use them again around tassel but no later when you may need the fungicide to finish the season. (See Dr. Telenko’s posts above).
3-Resistance management and integrated pest management. The photo below shows the economic threshold is 5-7% leaf severity for tar spot before it’s economical to spray. We’re a long way from that threshold on leaves confirmed for tar spot thus far. Avoiding unnecessary fungicide applications and using two modes of action when fungicides are applied may help in delaying resistance.
What to do now:
1-Continue to scout fields and wait till a 5-7% threshold on leaves before applying fungicide (see photo below)
2-Observe fields as to which hybrids have more tolerance to tar spot
3-When irrigating, consider less frequent and deeper irrigations
4-Consider plant nutrition? Manganese, Zinc, Copper, Boron, Sulfur all have a role in defending plants against pathogens. Will share more thoughts in another blog post.


Slide courtesy of Dr. Tamra Jackson-Ziems, UNL and Dr. Telenko, Purdue University. Fungicide application at V6-V7 resulted in negative economic return and similar disease pressure as the non-treated areas. Best fungicide timing for disease suppression occurred from tassel to milk and for economic return from tassel to dough.

Crop Updates 6/16/25
Wheat stem maggot in corn and herbicide injury issues have been my top questions the past few weeks. A team of us wrote a more detailed article of what we’re seeing and recommending regarding wheat stem maggot in CropWatch: https://go.unl.edu/atx9. Regarding herbicide injury, it perhaps is amazing this doesn’t happen more with all the traits we have. As spraying season continues, please double check fields for which traits are present to ensure the wrong herbicide isn’t applied to create potential crop injury. The one that has happened the most this season is glufosinate (Liberty) applied to non-Liberty link corn. The growth stage timing and rates of those applications make a huge difference on what we’ll see on corn survival.
Also, please always be checking growth stage of plants inside the field prior to herbicide applications. Crops have shorter internodes early on making them somewhat shorter than what we might expect. With June 21 coming this weekend as the longest day of the year, I’m anticipating we’re also going to see some blooms by this weekend/early next week with as many soybeans went in early this year.
As side-dress applications of nitrogen have been made in fields, it made me think of more on-farm research studies we’re conducting this year that might be of interest. They all involve using plant-based sensing from Sentinel Nutrient Management (also known as Sentinel Fertigation). Since not everyone who has irrigation systems wants to apply nitrogen through their system and because we have plenty of non-irrigated acres in the state, On-Farm Research is working with Sentinel to sense what the crop needed for side-dress applications this past year. There’s also opportunity for those with hi-boy machines to use this technology to apply nitrogen later in the season.
You’ve heard me share the on-farm research results about nitrogen savings using this sensing technology. In our area of the state, growers have used this technology in corn, soybean, and seed corn. This year, I know of growers also trying it in wheat and sorghum.
For on-farm research, I’m super excited to learn more from three studies that have had a history of cover crops and grazing! Each year, growers ask when to expect the nitrogen and other nutrients to be released from cover crops. Journal articles fairly consistently mention a 6-week time-frame. It ultimately depends on the Carbon:Nitrogen (C:N) ratio of the covers and rainfall to activate the biology. I’m excited to see if we can capture this time-frame better through satellite imagery using Sentinel Nutrient Management. The goal would be to see if we could better time nitrogen applications in addition to rates better using sensing technology.
Another question growers have asked is “how do we know how much nitrogen to credit after grazing cover crops?”. We hope to better answer this on two fields this year using sensing technology. Two of the on-farm research studies are following “Regen Years” where the land was planted to multi-species cover crop mixes and grazed all year instead of taking crops for grain. One is irrigated while the other is non-irrigated and both have nitrogen rate ramps associated with them. We plan to have a field day on the irrigated site in late July and will share more details later. Looking forward to what we will learn and sharing the results!
Reminder: June 25 Weed Management Field Day at UNL’s South Central Ag Lab near Clay Center, NE. Details at: https://go.unl.edu/qemk.




JenResources 6/8/25
Wheat Stem Maggot in Corn: Been seeing and hearing of some wheat stem maggot in corn for some fields where corn was planted green into rye. While I see this to a small extent each year, the last time we heard widespread reports was in 2017 (photos and info. at: https://jenreesources.com/2017/06/05/corn-concern-in-wheatrye-cover/). Symptoms include the corn whorls of plants looking wilted. Pulling out the whorl and carefully unfurling it may reveal the light-green maggot. Some key points: the maggot will move from dying rye or wheat into healthy corn. It will not move from plant to plant in corn unless the entire corn plant dies. What we have normally seen is the maggot kills the main stem of the corn plant; however, the plant produces tillers which then produce ears (not typical tiller ears). Because of that, replanting is not recommended. Our Extension Entomologists also don’t recommend spraying an insecticide once the maggots are in the whorls because they won’t have activity against the maggots inside the corn. There are no published thresholds for the maggot once it gets into the corn plant. Here’s an article UNL researchers published on timing and use of insecticides in cover crop/corn systems: https://www.mdpi.com/2075-4450/13/4/348.
Regen Ag Field Tours: A series of Regen Ag tours are available June-August. For now, I’ll share on two of them. On June 11, Doug and Traci Steffen are hosting a compost and covers tour on their farm near Crofton, NE from 1-3 p.m. RSVP at https://practicalfarmers.org/field-days.
On July 8 from 10 a.m.-Noon, Junior Pfanstiel is hosting a covers and corn tour on his farm near McLean, NE. Lunch is also included. Junior alternated 4 rows of corn with 4 rows of multi-species cover crops throughout his field with the goal of obtaining the same yield on fewer acres while also having grazing acres. Please RSVP to: (402) 254-6758.
Roundup: Homeowners, Please read the active ingredients on jugs before purchasing and using Roundup products. I’ve had several situations where homeowners have bought “Roundup” thinking it was the traditional “glyphosate”, but instead the active ingredient was “triclopyr”. The problem? They work differently. Glyphosate is a non-selective herbicide that doesn’t move in the soil or from plant to plant. It only works on the plant it comes in contact with. Triclopyr is a growth regulator herbicide (same family as 2,4-D and dicamba) that mostly affects shrubs, stump treatment, and broadleaf weeds. It has minimal activity on grasses. It has soil residual activity, on average for 30 days, but can be as much as nearly a year depending on how the microbial life break it down. It can volatilize (move off-target to other broadleaf plants after application) and it can be excreted through the roots of plants that were sprayed to nearby, non-sprayed plants impacting them.
Because of this, some people who have used Roundup containing triclopyr have reported issues with growing vegetables, flowers, their grape vines, raspberries, and around fruit trees. Triclopyr symptoms on plants looks like a “strapping” of leaves where they become bubbly and elongated, but it’s a different look than 2,4-D damage. Plants grown in soil treated with triclopyr may have kinked stems and appear stunted. More info. on triclopyr: https://purduelandscapereport.org/article/the-summer-of-triclopyr-mounting-evidence-for-off-target-damage/. A great resource explaining all the different “Roundup” products on the market: https://www.purdue.edu/fnr/extension/when-roundup-isnt-roundup-purdue-landscape-report/.








